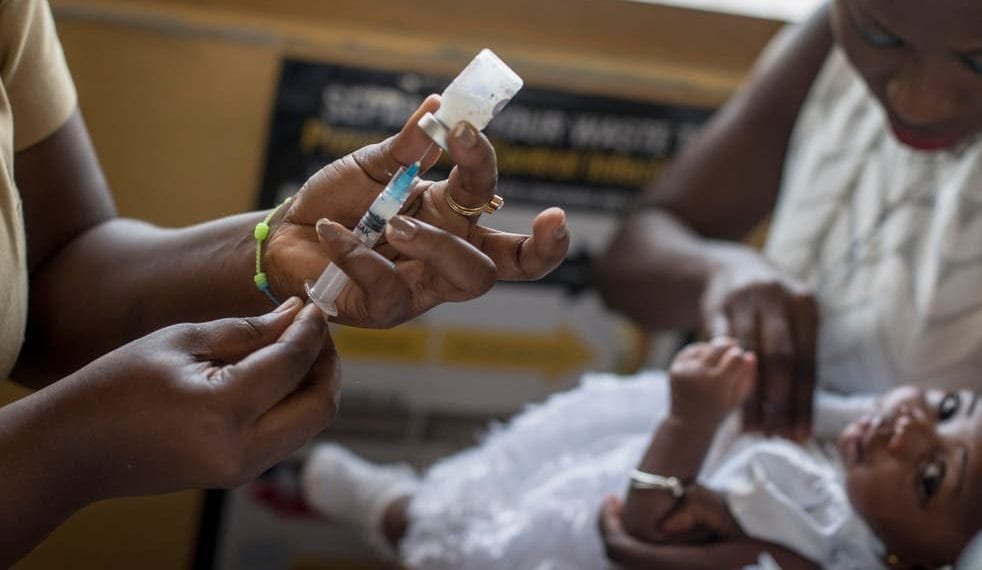
Researchers from the Oxford University have developed the “world’s most effective malaria vaccine”,;with the intention of slashing the death toll of a disease that kills 400,000 people, mostly small children, every year.
The vaccine, developed by scientists at the Jenner Institute of Oxford University,;proved to 77% efficient in a trial of 450 children in Burkina Faso over 12 months.
It is also the first to meet the World Health Organization’s (WHO) goal of 75% efficacy against malaria. Scientists say larger trials are now beginning, involving 4,800 children in four countries.
In an interview, Professor Adrian Hill, Director of the Jenner Institute, said he believed the vaccine had;the potential to cut the death toll dramatically.
“What we’re hoping to do is take that 400,000 down to tens of thousands in the next five years,;which would be absolutely fantastic.”
He noted that other interventions, such as impregnated bednets and malarial drugs, have reduced;the death toll from a million a year and agreed that those initiatives must continue. However, he indicated that if the vaccine could cut deaths to tens of thousands,;the institute might be able to look towards “a greater goal of eventually eradicating malaria”.

Hill also said the institute might apply for emergency approval for the malaria vaccine;just as it did for the Oxford/AstraZeneca Covid jab.
“I’m making the argument as forcefully as I can, that because malaria kills a lot more people than Covid in Africa, you should think about emergency-use authorization for a malaria vaccine for use in Africa. And that’s never been done before.
“They did Covid in months – why shouldn’t they do malaria in a similar length of time as the health problem is an even greater scale in Africa?”
End of 2022 best-case scenario for approval
The researchers have also indicated that the vaccine will be manufactured at a large scale and low-cost after a deal with the Serum Institute of India. The Serum Institute is also involved in manufacturing of the Oxford/AstraZeneca Covid-19 vaccine.
The Institute has had to delay supplies of the Covid vaccine to the rest of the world because of the huge rise in cases in India, but has promised to deliver 200m doses a year of the malaria vaccine if it is licensed.
Professor Hill noted that the best-case scenario was approval by the end of 2022, by which time the Serum Institute would have plenty of capacity.
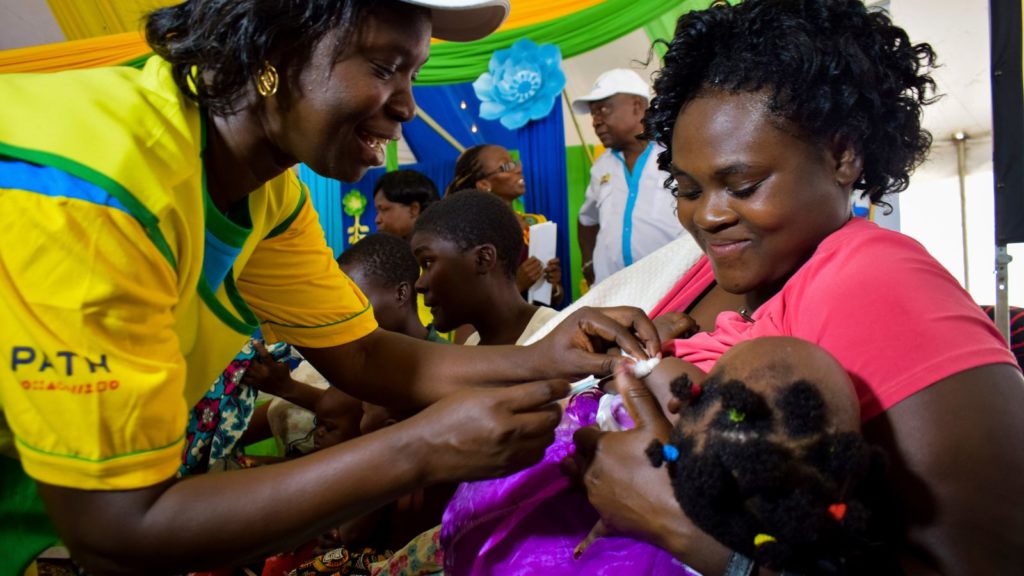
In a statement, chair and CEO of the Serum Institute, Dr Cyrus Poonawalla and Adar Poonawalla, indicated that they were “highly excited to see these results on a safe and highly effective malaria vaccine which will be available to the whole world”.
“We are highly confident that we will be able to deliver more than 200m doses annually in line with the strategy, as soon as regulatory approvals are available”.
The search for a malaria vaccine has been going on for a long time. One, the Mosquirix vaccine developed by GlaxoSmithKline, has been through lengthy clinical trials but is only partially effective. It prevents 39% of malaria cases and 29% of severe malaria cases among small children in Africa over four years. It is being piloted by the WHO in parts of Kenya, Ghana and Malawi.
Read Also: GPP holds a mentorship and business boot camp for young entrepreneurs